New Dental Hygiene Technology In 2026
Dental hygiene moved through a noticeable inflection point in early 2026. Within roughly four months, a major water flosser line received an automated intensity-control upgrade,...
Written by Marcus Hale
Read time: 8 min read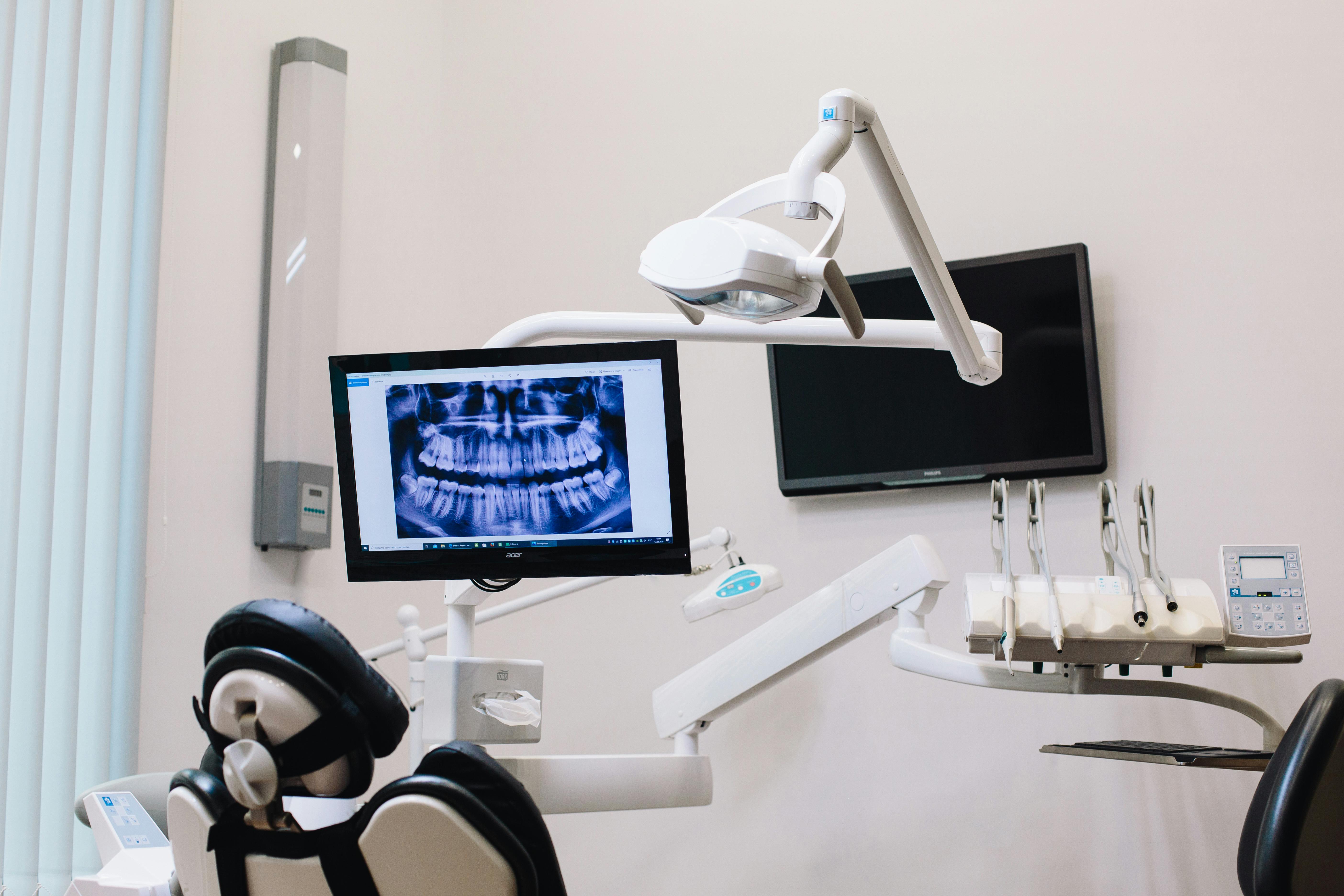
Dental hygiene moved through a noticeable inflection point in early 2026. Within roughly four months, a major water flosser line received an automated intensity-control upgrade, the largest dental AI vendor in the United States expanded its FDA-cleared scope to panoramic radiographs, and a peer-reviewed narrative review took stock of every standalone AI imaging product cleared by the FDA so far. These shifts touch the at-home routine, the operatory chairside experience, and the way a hygienist documents and discusses findings with a patient.
This article covers the dental hygiene technology that was actually released or meaningfully upgraded in 2026. Older platforms still in clinical use are referenced for context, but the focus stays on what is new this year. Sections move from consumer-facing devices to the in-office layer, with practical notes for clinicians and the patients they care for.
Why 2026 Is A Pivotal Year For Dental Hygiene Technology
A combination of consumer expectation, regulatory progress, and clinical evidence pushed the category forward this year. Three forces explain most of the momentum, and recognizing them helps clinicians evaluate any new product against a useful baseline.
The first is the maturation of FDA-cleared dental AI. According to a 2026 narrative review published in the International Dental Journal, thirteen companies offer twenty-nine FDA-cleared AI products for dental imaging across caries detection, periodontal assessment, cephalometric analysis, multi-pathology diagnostics, automated charting, and three-dimensional segmentation. Clearance dates cluster between 2022 and 2025, with continued expansion into 2026 as platforms add new indications.
The second force is consumer expectation. People who track sleep, steps, and heart rate now expect their oral care devices to track and adapt as well. A water flosser without progressive intensity, a toothbrush without quadrant timing, or a charting workflow without AI assistance is increasingly a generation behind.
The third force is the unmet clinical need. According to CDC oral health surveillance data, roughly one in four US adults aged twenty to sixty-four has untreated tooth decay. Daily home care remains the highest-leverage input on caries and gum disease outcomes. Any home-care upgrade with credible adherence benefits has a real public-health argument behind it.
The Waterpik ProMax With SmartAdvance, The Biggest Consumer Launch Of 2026
The most prominent at-home oral care launch of 2026 came from Waterpik. On March 20, 2026, World Oral Health Day, the brand introduced the ProMax water flosser with SmartAdvance mode, a countertop model designed to remove the most common reason patients abandon oral irrigation, namely discomfort with water pressure on day one.
The clinical relevance of this launch lies in patient adherence rather than raw mechanical power. ProMax solves a behavioral problem that string floss has never solved well.
What SmartAdvance Mode Actually Does
SmartAdvance is an automated pressure-progression mode rather than a connected app feature. The implementation is straightforward, and its value lies in what it removes from the user's mental load.
According to the Waterpik product information, SmartAdvance starts a new user at intensity level 3 and gradually increases to level 9 or higher over two weeks of daily use. Waterpik states that consistent use can help reverse bleeding gums in as little as six weeks when paired with the Precision tip. The device is ADA-accepted and uses Waterpik's PrecisionPulse technology for combined water pressure and pulsation.
Independent coverage from DrBicuspid and Compendium confirms the same feature set, including QuietPulse noise control, dual-user memory, and five specialty tips. CNN Underscored's product launch coverage lists the device at $149.99 across major retailers.
For dental professionals working with patients new to water flossing, SmartAdvance reduces a familiar conversation. Where clinicians previously had to coach patients on stepping up intensity manually, the device handles that progression on its own. For patients with sensitive gums, recent gingivitis diagnoses, or a history of abandoning oral irrigation devices, ProMax is worth a specific recommendation.
Where ProMax Fits Versus Earlier Waterpik Models
ProMax sits at the top of the Waterpik countertop lineup, above the long-running Aquarius and similar countertop models. The differentiation is the automated intensity control, the noise reduction, and the dual-user memory rather than a meaningful change in cleaning physics.
For independent comparison and ranking against competing oral irrigators, the Dental Reviewed best oral irrigator roundup for 2026 provides a current overview. Patients with braces, implants, or active periodontal disease should pay particular attention to tip variety and pressure ceiling rather than focusing on the smart features alone.
AI In Dental Hygiene, 2026 Updates To FDA-Cleared Imaging Platforms
The clinical AI landscape shifted in early 2026 with new FDA clearances and a comprehensive review of the category. For hygienists and dentists, these updates change the operating environment of a routine recall visit, particularly the radiograph review and the patient education portions.
The most important updates came from Pearl, a continued expansion of Overjet's clinical footprint, and the narrative review that mapped the entire FDA-cleared field.
Pearl Second Opinion's Panoramic Radiograph Clearance
Pearl's most significant 2026 milestone was an expanded FDA clearance covering panoramic radiographs.
In early 2026, Pearl announced FDA 510(k) clearance for AI-assisted detection of dental pathologies on panoramic radiographs. The cleared functionality identifies suspected caries, periapical radiolucencies, and impacted third molars, and is now available to Second Opinion users in the United States and internationally.
The clinical significance lies in modality coverage. Panoramic imaging is the most commonly captured extraoral radiograph in dentistry, and earlier AI clearances focused predominantly on bitewing and periapical views. Bringing AI overlay support to panoramic films extends consistent diagnostic assistance to a workflow where it was previously absent.
For an independent perspective on Pearl's platform value, the Dental Reviewed Pearl AI review walks through the practical case in detail.
The 2026 FDA-Cleared AI Landscape At A Glance
Catalogs of the FDA-cleared dental AI category have been scarce, which made the publication of a peer-reviewed mapping of the field a quiet but important development this year.
The most useful single reference for clinicians evaluating AI adoption this year is the narrative review published in the International Dental Journal. The review cataloged thirteen companies offering twenty-nine FDA-cleared AI imaging products and analyzed the supporting peer-reviewed evidence behind each.
Two findings stand out. Overjet leads the field with nine FDA-cleared modules covering caries detection, calculus detection, periapical radiolucency, automated charting, and image enhancement. Pearl follows with seven cleared modules across bitewing, periapical, panoramic, and CBCT modalities. Independent peer-reviewed validation varies meaningfully across vendors, with Relu Creator and WebCeph supported by the highest publication counts in the review's analysis.
For clinicians integrating AI into the hygiene visit, three practical implications follow. AI overlays improve diagnostic consistency between team members reviewing the same image. Patients are more likely to accept treatment recommendations when the finding is visualized on their own radiograph. Bone-loss measurement gains substantial inter-operator reliability when an AI tool produces the measurement rather than a hygienist doing it visually.
A useful starting point for non-clinician readers exploring this category is the primer on AI in dentistry, which covers the practical applications and limits in plain terms.
How AI Improves The Hygiene Visit Workflow
The downstream effect of FDA-cleared AI on a recall appointment is more than a screen overlay. It changes the documentation, the patient conversation, and the consistency of treatment planning across visits.
Modern AI platforms change three concrete steps during a recall appointment. The hygienist reviews radiographs alongside an AI overlay that highlights candidate findings, the dentist's exam confirms or revises those findings against clinical judgment, and the resulting dental treatment plan reflects a baseline that is more consistent across visits and across team members.
Two limits should not be glossed over. AI is decision-support, not autonomous diagnosis, so the dentist still owns the final call. FDA clearance applies to specific indications, not general-purpose interpretation, and clinicians should verify what a clearance covers before relying on a tool for a given task.
Smart Toothbrush Refinements And Subscription Models In 2026
Smart toothbrush hardware has been relatively stable, with most premium models from Philips Sonicare and Oral-B continuing to lead on sensor quality and app integration. The 2026 development worth noting is the broader normalization of subscription-delivered replacement heads, dual-user memory, and pressure feedback as baseline expectations rather than premium add-ons.
For patients evaluating the category, the practical features that matter remain consistent.
The ADA Seal of Acceptance, which confirms an independent evaluation for safety and efficacy
A two-minute timer with thirty-second quadrant pacing
A pressure sensor visible on the handle, ideally also surfaced in the app
Replaceable heads on a clear three to four-month schedule
Battery life of at least two weeks under normal use
Reliable subscription delivery if auto-replacement matters to the user
A travel case with USB-C charging for frequent travelers
Independent reviews continue to be the most reliable starting point for individual model selection. The guide on smart toothbrush use covers the clinical rationale, and the roundup of electric toothbrushes for receding gums addresses one of the most common patient profiles in 2026.
New 3D Printing Capabilities Affecting Hygiene-Adjacent Workflows
3D printing technology used in restorative dentistry continued to mature in 2026, with implications for the hygienist who later maintains those restorations. The most notable advance changes the timeline for in-office zirconia fabrication.
According to coverage from Open Dental, researchers at the University of Texas at Dallas announced a 3D printing process in late 2025 that reduces zirconia debinding time from weeks to under thirty minutes, opening the possibility of in-office zirconia crowns, veneers, and bridges in routine workflows.
The clinical relevance of hygiene is twofold. Patients arriving for recall increasingly present with same-day or near-same-day milled or printed restorations rather than lab-fabricated ones, which changes the maintenance conversation. Zirconia surfaces respond to ultrasonic instrumentation, polishing pastes, and air polishing differently than ceramic and metal, so hygienists working with patients who received printed zirconia in 2026 should adjust their instrument selection accordingly.
For a broader context on dental laser use in hygiene-adjacent procedures, the dental lasers reference covers wavelength categories, indications, and certification expectations.
Cloud-Native Practice Management As A 2026 Standard
The 2026 dental practice technology landscape shifted decisively toward cloud-native practice management platforms, a transition with practical implications for hygiene teams adopting AI-cleared tools.
According to a HealthStream Ventures report, cloud-native systems now provide measurable operational advantages over legacy server-based systems, including automation, workflow consistency, AI readiness, and improved visibility across clinical, financial, and patient engagement workflows.
The implication for hygiene operations is direct. Cloud-native platforms allow AI-cleared imaging tools, voice-driven periodontal charting, and automated recall communication to integrate without on-premise server upgrades. Practices that adopted cloud platforms before 2026 are positioned to add AI-enabled tooling more efficiently than practices still running legacy systems.
For hygienists and assistants evaluating new platforms, the primer on dental imaging AI and the coverage of AI governance in dentistry together provide a current professional baseline.
What These 2026 Releases Mean For Patients And Clinicians
Three patient profiles cover most of what this year's launches and clearances mean in practice. A clean recommendation falls out of each.
Adults with bleeding gums or new gingivitis diagnoses are the most direct beneficiaries of the Waterpik ProMax launch. The automated intensity progression removes the most common reason this group abandons oral irrigation in the first month.
Adults attending a recall in a practice that uses an FDA-cleared AI platform should expect to see their radiographs reviewed alongside an AI overlay, which improves diagnostic consistency and supports a clearer conversation about treatment recommendations. For an independent context on smart oral care wearables more broadly, the dental wearables explainer covers the connected-device landscape.
Patients with new restorations, particularly zirconia crowns or bridges produced through emerging 3D printing workflows, should expect their hygiene team to adjust instrument selection during cleanings.
Bottom Line
The 2026 dental hygiene technology cycle is best understood as the year automation displaced manual judgment in three specific places. Water flosser intensity progression became automated through SmartAdvance, panoramic radiograph review gained an FDA-cleared AI overlay through Pearl's expanded clearance, and the broader FDA-cleared AI imaging field reached twenty-nine cleared products across thirteen vendors as cataloged in the International Dental Journal review.
None of this replaces the basics. Brushing twice for two minutes, cleaning between teeth daily, and attending two recall visits per year remain the foundation. Technology earns its place when it makes the basics easier to maintain and easier to verify, not when it claims to bypass them. Read the labels, check the ADA Seal database, ask the dentist what they actually use in the office, and treat any product claim that sounds dramatic with the skepticism appropriate to any health-tech category.
Frequently Asked Questions
What is the most important new dental hygiene technology released in 2026?
The Waterpik ProMax with SmartAdvance mode, launched on March 20, 2026, is the most consumer-relevant release of the year. On the clinical side, Pearl's FDA clearance for AI-assisted pathology detection on panoramic radiographs is the most significant regulatory expansion.
How does the Waterpik ProMax differ from earlier Waterpik models?
ProMax adds SmartAdvance, an automated pressure progression that takes new users from level 3 to level 9 or higher over two weeks. It also includes QuietPulse noise reduction, dual-user memory, and five specialty tips, and it is ADA-accepted.
What FDA-cleared AI tools are available for dental hygiene visits in 2026?
A 2026 narrative review in the International Dental Journal identified twenty-nine FDA-cleared AI products from thirteen companies, covering caries detection, periodontal assessment, cephalometric analysis, multi-pathology diagnostics, automated charting, and three-dimensional segmentation. Overjet leads with nine cleared modules, and Pearl follows with seven.
Are AI-powered diagnostic tools replacing dentists in 2026?
No. FDA-cleared dental AI is decision-support software. The dentist still owns the diagnostic decision, and clearance applies only to specific indications, not general-purpose interpretation.
Should patients with sensitive gums use the Waterpik ProMax?
Yes, patients with sensitive gums or recent gingivitis are the most direct beneficiaries of the SmartAdvance mode. The gradual pressure progression is designed for users who would otherwise abandon oral irrigation due to first-day discomfort.
What is changing in 3D printed restorations in 2026?
A 3D printing process announced by University of Texas at Dallas researchers in late 2025 reduces zirconia debinding time from weeks to under thirty minutes. As the process moves toward broader clinical use, hygienists will encounter more in-office zirconia restorations during recall visits and should adjust their instrumentation accordingly.
Why do practices need cloud-native software in 2026?
Cloud-native platforms enable easier integration with FDA-cleared AI imaging tools, voice-driven periodontal charting, and automated patient communication. Legacy server-based systems often require infrastructure upgrades to support these workflows.
How can patients verify that a smart oral care product has been independently evaluated?
Check the ADA Seal of Acceptance product list, which confirms a specific model has been independently evaluated for safety and effectiveness. ADA acceptance is voluntary, so its absence does not condemn a product, but its presence is a meaningful independent signal.
Where can dental professionals build current treatment plans efficiently?
AI-assisted treatment planning platforms have matured into a real category in 2026. Tools like the Dental Reviewed AI dental treatment plan generator produce evidence-based plans in seconds and remain fully editable by the clinician.