Infection Control in Dentistry: Complete Guide (2026)
Effective dental infection control isn't just about regulatory compliance. It's about creating an environment where patients receive care with confidence and staff work safely....
Written by Mantas Petraitis
Read time: 11 min read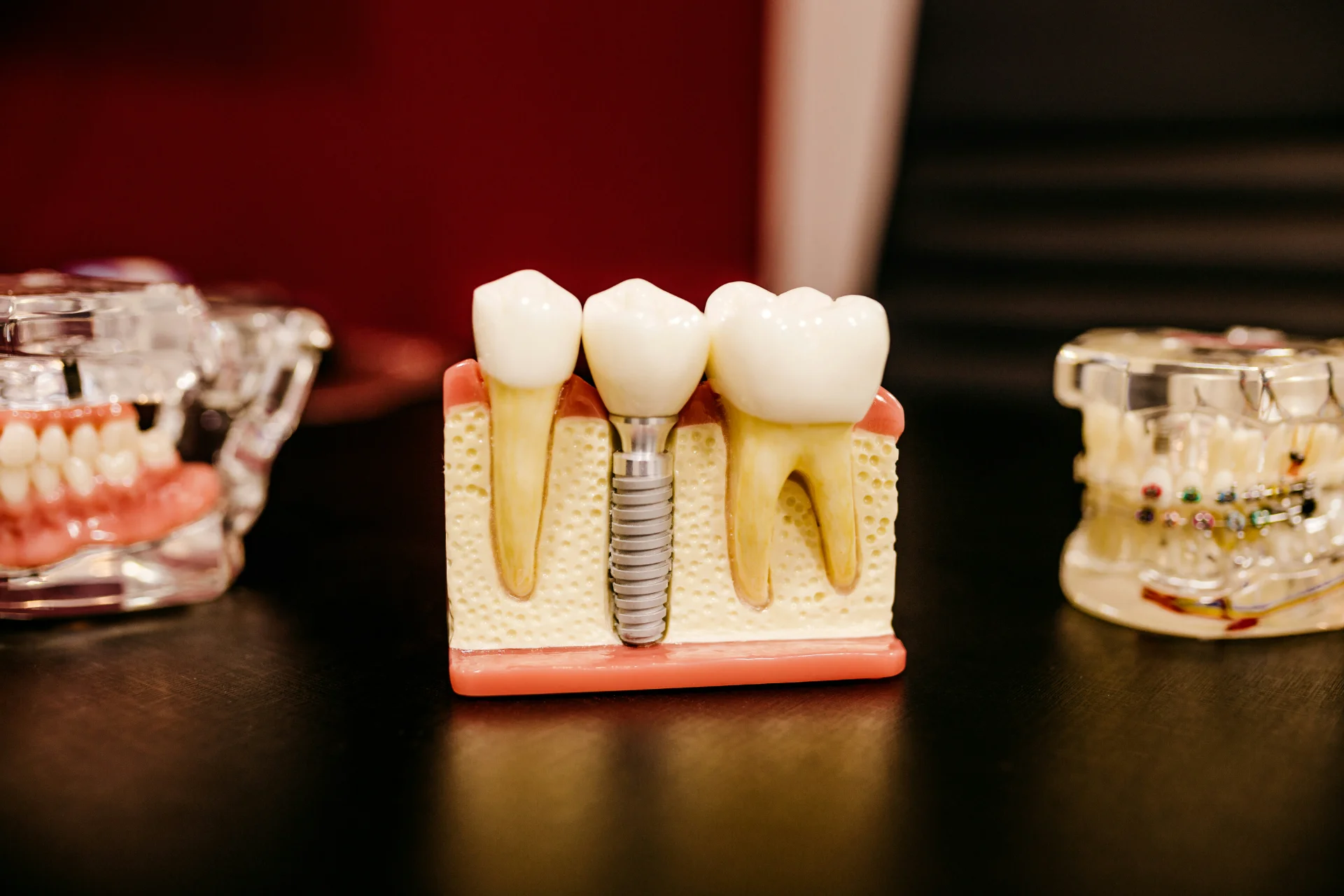
Effective dental infection control isn't just about regulatory compliance. It's about creating an environment where patients receive care with confidence and staff work safely. This comprehensive guide covers the essential protocols, regulatory requirements, and best practices that form the foundation of infection control in modern dental practice. Whether you're a dental professional seeking to update your protocols, a practice manager ensuring compliance, or a patient wanting to understand what safety measures should be in place, this guide provides the authoritative information you need.
Regulatory Framework and Guidelines
Understanding the regulatory landscape is essential for implementing compliant and effective infection control protocols. Multiple agencies provide overlapping guidance that creates a comprehensive framework for dental infection control.
The Centers for Disease Control and Prevention (CDC) published comprehensive Guidelines for Infection Control in Dental Health-Care Settings in 2003, with important updates in 2016 and additional interim guidance during the COVID-19 pandemic. These guidelines represent the gold standard for infection prevention in dentistry, covering everything from hand hygiene to instrument processing. The CDC takes an evidence-based approach, categorizing recommendations by the strength of supporting scientific data. Dental practices should regularly consult the CDC website for the most current guidance, as recommendations evolve based on emerging research and disease threats.
The Occupational Safety and Health Administration (OSHA) enforces the Bloodborne Pathogens Standard (29 CFR 1910.1030), which carries the force of law. This regulation requires dental practices to develop and maintain a written Exposure Control Plan, provide hepatitis B vaccinations to at-risk employees, use appropriate personal protective equipment, implement engineering and work practice controls, and maintain detailed training and incident records. Unlike CDC guidelines, which are recommendations, OSHA standards are mandatory and enforceable through inspections and citations. Failure to comply can result in significant fines and legal liability.
The Food and Drug Administration (FDA) regulates dental equipment and devices, including sterilization equipment, dental unit waterlines, and single-use devices. The FDA classifies medical devices by risk level and requires appropriate clearance before products can be marketed. This regulatory oversight ensures that sterilization equipment meets performance standards and that manufacturers provide validated instructions for use. Dental practices must follow FDA-cleared procedures and cannot arbitrarily modify device processing protocols.
State dental boards often impose additional requirements beyond federal regulations. Some states mandate more frequent biological indicator testing, require specific sterilization methods, or have unique documentation requirements. It's essential to know your state's specific regulations, which are typically available through your state dental board website. International practices should consult WHO guidelines and local regulatory bodies, as requirements vary significantly by country.
While many countries have created their own regulatory framework, the essentials covered by the main, world-renowned health agencies remain crucial for every practitioner.
Fundamental Concepts and Terminology
Before implementing specific protocols, understanding core infection control concepts provides the foundation for effective practice. These fundamental principles guide decision-making and help staff understand the rationale behind procedures rather than simply following steps by rote.
Standard Precautions
Standard precautions represent the foundation of infection control in all healthcare settings. This approach assumes that all patients are potentially infectious for bloodborne pathogens and other transmissible diseases, regardless of diagnosis or perceived infection status. Standard precautions include hand hygiene, use of personal protective equipment based on anticipated exposure, respiratory hygiene and cough etiquette, safe injection practices, and proper handling of contaminated equipment and surfaces. This concept evolved from the earlier "universal precautions" terminology but broadened the scope to include all body fluids, not just blood. By treating every patient encounter as if infectious agents could be present, dental professionals eliminate the risk of making incorrect assumptions about patient infection status.
Spaulding Classification
The Spaulding Classification system, developed in 1968 but still relevant today, categorizes medical and dental instruments based on infection risk and determines appropriate processing methods. Critical instruments penetrate soft tissue or bone and carry the highest risk of infection transmission. These items, such as surgical instruments, scalers, and burs, must be sterilized between each use. Semi-critical instruments contact mucous membranes or non-intact skin but don't penetrate tissue. Examples include mirrors, impression trays, and condensers. These items require, at a minimum, high-level disinfection, though heat sterilization is preferred when possible. Non-critical items contact only intact skin and require low-level to intermediate-level disinfection. Understanding this classification helps dental teams make appropriate processing decisions and allocate resources effectively.
Chain of Infection
The chain of infection concept explains how diseases spread and identifies intervention points. Six links must be present for infection transmission: an infectious agent (bacteria, virus, fungi), a reservoir where the agent lives and multiplies (human, equipment, environment), a portal of exit from the reservoir (blood, saliva, respiratory droplets), a mode of transmission (direct contact, droplet, airborne), a portal of entry into a new host (mucous membranes, broken skin, inhalation), and a susceptible host who lacks immunity. Effective infection control breaks this chain at multiple points. For example, hand hygiene prevents transmission, PPE blocks portals of entry, and sterilization eliminates infectious agents. Understanding this framework helps dental professionals see how various protocols work together as an integrated system rather than isolated procedures.
Levels of Disinfection and Sterilization
Disinfection and sterilization represent different levels of microbial destruction. Sterilization destroys all forms of microbial life, including bacterial spores, and is the only acceptable processing for critical instruments. High-level disinfection eliminates all microorganisms except high numbers of bacterial spores and is appropriate for semi-critical items when sterilization isn't feasible. Intermediate-level disinfection kills mycobacteria, vegetative bacteria, most viruses, and fungi but doesn't kill bacterial spores. This level is appropriate for environmental surfaces in dental settings. Low-level disinfection kills most vegetative bacteria, some fungi, and enveloped viruses but not mycobacteria or spores. This is adequate for housekeeping surfaces with minimal hand contact. Understanding these distinctions ensures appropriate processing methods are selected for different applications.
Personal Protective Equipment (PPE)
Personal protective equipment serves as the first line of defense protecting dental healthcare personnel from exposure to infectious materials. Proper selection, use, and disposal of PPE are critical components of any infection control program.
Gloves
Gloves create a barrier between hands and potentially infectious materials, but only when used correctly. Medical exam gloves are appropriate for most patient care activities and should be changed between patients, when torn or punctured, and whenever leaving the patient care area. Surgical gloves provide higher tactile sensitivity and a better fit for surgical procedures. Utility gloves, which are thicker and reusable, protect hands during instrument cleaning and surface disinfection. Never wash or disinfect patient care gloves for reuse, as this damages the material and increases permeability. Staff with latex allergies should use nitrile or vinyl alternatives. A common mistake is wearing the same gloves for multiple tasks. Gloves contaminated during patient care should never touch charts, phones, or doorknobs outside the treatment area.
Masks and Respirators
Understanding the difference between surgical masks and respirators is crucial, especially following COVID-19. Surgical masks protect others from the wearer by containing large respiratory droplets and sprays. They're rated by ASTM levels (Level 1, 2, or 3) based on fluid resistance and bacterial filtration efficiency. However, surgical masks don't seal tightly to the face and don't filter small airborne particles effectively. N95 respirators, in contrast, filter at least 95% of airborne particles and are required for aerosol-generating procedures or when treating patients with suspected airborne diseases. N95s require fit testing to ensure proper seal, and users should perform a seal check each time they don the respirator. Masks and respirators should be changed when they become wet, contaminated with blood or body fluids, or at least between patients. Never allow masks to dangle around the neck, as this contaminates both the mask and the wearer.
Eye Protection and Face Shields
Eye protection is mandatory whenever splashes, sprays, or aerosols might be generated. Protective eyewear must have solid side shields to protect against exposure from the side. Personal prescription glasses alone provide inadequate protection and don't meet OSHA requirements. Face shields provide additional protection, covering the entire face and protecting areas that eyewear alone misses. During COVID-19, face shields became standard in combination with masks and eye protection to provide maximum barrier protection. Between patients, face shields should be cleaned and disinfected according to manufacturer instructions, typically using an EPA-registered hospital disinfectant. Anti-fog features help maintain visibility during procedures.
Protective Clothing
Clinical attire should have long sleeves to protect forearms from contamination. Fluid-resistant gowns or jackets are preferred, especially for procedures likely to generate splash or spray. Lab coats worn in clinical areas should be removed before leaving the practice and laundered at the facility or by a professional service, never taken home. Change protective clothing when visibly soiled or contaminated with blood or body fluids. Some practices use disposable gowns that are discarded after each patient, eliminating laundry concerns and ensuring fresh barriers for every patient encounter.
Proper Donning and Doffing Sequence
The sequence in which PPE is put on and removed matters significantly for preventing self-contamination. When donning PPE, start with the gown, then the mask or respirator, followed by eye protection, and finally gloves. This sequence ensures each item is properly positioned. Doffing requires even more care to avoid contamination. Remove gloves first using a technique that prevents hand contact with the contaminated exterior. Perform hand hygiene immediately after glove removal. Remove eye protection by grasping the clean headband or earpieces. Remove the gown by rolling it away from the body so the contaminated outside is turned inward. Finally, remove the mask or respirator by touching only the ties or straps, never the front. Perform hand hygiene again after all PPE removal. Many practices post visual aids showing proper sequences to reinforce correct technique.
Hand Hygiene
Hand hygiene stands as the single most important measure for preventing disease transmission in healthcare settings, yet compliance remains a persistent challenge in many practices.
Dental professionals must perform hand hygiene at specific critical moments: before donning gloves and after removing them, before and after patient contact, after touching contaminated surfaces or equipment, before eating or handling food, after restroom use, and whenever hands become visibly soiled. The "before donning gloves" step often surprises people, but contaminated hands can lead to glove contamination from the inside, and gloves can develop micro-tears during use that allow contact with hands.
When hands are visibly soiled or contaminated with blood or body fluids, handwashing with soap and water is required. Proper handwashing technique involves wetting hands with water, applying enough soap to cover all surfaces, rubbing hands together to create friction for at least 20 seconds, covering all surfaces, including backs of hands, between fingers, and under nails, rinsing thoroughly under running water, and drying completely with a disposable paper towel. Use the paper towel to turn off the faucet to avoid recontaminating clean hands. Water temperature is less critical than duration and friction. Lukewarm water is comfortable and just as effective as hot water.
For most routine clinical situations where hands aren't visibly soiled, alcohol-based hand rubs offer significant advantages over soap and water. They're faster, more convenient, less irritating to the skin, and actually more effective at reducing bacterial counts. Apply enough product (usually one palm-full) to cover all hand surfaces, rub hands together covering all surfaces until hands are completely dry, which typically takes 20-30 seconds. The drying process is essential for effectiveness – don't wipe hands or rinse off the product. Hand rubs should contain 60-95% alcohol for optimal antimicrobial activity. However, alcohol-based hand rubs aren't effective against certain pathogens like C. difficile spores, norovirus, and when hands are visibly soiled, making soap and water necessary in these situations.
Nail care represents an often-overlooked aspect of hand hygiene. Natural nails should be kept short, typically no more than ¼ inch beyond the fingertip. Long nails harbor more microorganisms, are more likely to puncture gloves, and interfere with thorough hand cleaning. Artificial nails, including gels, acrylics, wraps, and tips, are strongly discouraged or prohibited in most dental settings, as they're associated with increased bacterial counts and have been linked to disease outbreaks in healthcare facilities. Nail polish, if worn, should be fresh and chip-free, as chipped polish can harbor bacteria. Rings and bracelets should be removed or minimized, as they interfere with effective handwashing and harbor microorganisms.
Finally, hand care itself matters for infection control. Damaged skin with cuts, cracks, or dermatitis has impaired barrier function and increased risk of infection. Encourage staff to use moisturizers regularly to maintain skin integrity, avoid harsh soaps that overdry skin, cover any cuts or abrasions with waterproof bandages before donning gloves, and seek medical attention for persistent skin problems. Healthy, intact skin is an essential component of personal protection.
Instrument Processing: Overview and Workflow
Proper instrument processing follows a systematic workflow designed to move items from contaminated to clean to sterile while preventing cross-contamination at each step.
Processing begins at the point of use in the operatory. Immediately after use, instruments should be kept moist to prevent blood and debris from drying, which makes cleaning much more difficult. Use a pre-cleaning foam or spray, or submerge instruments in water or an enzymatic solution. Never leave instruments exposed on the counter where they create aerosols and contamination. Transport instruments to the processing area in a closed, leak-proof container. Handle sharps with extreme care, using mechanical means rather than hands whenever possible. Never reach into containers to retrieve instruments, pour them into the sink or onto the work surface.
The central processing area should be designed with distinct zones to prevent cross-contamination. The receiving/dirty area is where contaminated instruments arrive and undergo cleaning. This area should be physically separated from clean areas and have negative air pressure if possible. The preparation and packaging area is where cleaned instruments are inspected, assembled into sets, and packaged for sterilization. The sterilization area contains autoclaves and other sterilization equipment. Finally, the sterile storage area houses processed instruments until they're needed. Workflow should move in one direction only: from dirty to clean to sterile, never backward. Staff should understand they cannot move from the dirty area to the clean area without removing contaminated PPE and performing hand hygiene.
An adequate instrumentation inventory is essential for efficient processing. Practices need enough instruments to allow for proper cleaning, sterilization, and cooling times without shortcuts. Rush sterilization (unwrapped or immediate use steam sterilization) should be reserved for genuine emergencies only, not routine workflow problems. Overnight processing of instruments is acceptable as long as proper protocols are followed and dried instruments are packaged appropriately.
Documentation provides the audit trail essential for quality assurance and regulatory compliance. Sterilization logs should record date, load contents, cycle parameters, operator initials, and monitoring results. Biological indicator results must be documented and retained according to state requirements, typically 3-10 years. Equipment maintenance records demonstrate ongoing equipment performance. Training documentation shows staff competency. This documentation proves compliance during inspections and provides valuable quality assurance data for continuous improvement.
Cleaning of Dental Instruments
Cleaning is the most critical step in instrument processing, yet it's often underappreciated. No matter how effective your sterilization equipment is, you cannot sterilize a dirty instrument. Organic material, like blood and tissue, creates a barrier that prevents sterilants from contacting instrument surfaces, and dried debris is extremely difficult to remove.
Manual cleaning remains appropriate for delicate instruments that might be damaged by mechanical cleaning. When cleaning manually, wear heavy-duty utility gloves, protective eyewear, a mask, and a gown to protect against splashes. Use a long-handled brush to scrub instruments under the water line to minimize aerosol generation. Never scrub above water where splashing and aerosols occur. Use enzymatic or detergent cleaners according to the manufacturer's instructions. Pay special attention to hinges, serrations, and other areas where debris can hide. Rinse thoroughly to remove all detergent residue, which can interfere with sterilization and cause instrument damage. Manual cleaning is labor-intensive and exposes staff to contaminated instruments, so mechanical cleaning is preferred when possible.
Ultrasonic cleaners use high-frequency sound waves to create millions of microscopic bubbles in a cleaning solution, a process called cavitation. As these bubbles collapse, they create intense scrubbing action that reaches into crevices and removes debris more effectively than manual scrubbing. Load instruments with hinges open and arrange them so they don't touch each other or the basket sides, allowing the solution to contact all surfaces. Use the appropriate cleaning solution at the manufacturer-recommended concentration and temperature. Most cycles run 10-15 minutes, though heavily soiled instruments may require longer. After the cycle completes, rinse instruments thoroughly to remove loosened debris and cleaning solution residue. Change ultrasonic solution when visibly soiled, at least daily, and whenever the solution becomes diluted or contaminated. Before first use each day, degas fresh solution by running an empty cycle to remove dissolved air that interferes with cavitation. Don't clean delicate items like plastic instruments or fiber optic handpieces in ultrasonic cleaners, as the intense action can damage them.
Automated instrument washers represent the gold standard for instrument cleaning, offering consistent, reproducible results with minimal staff exposure to contaminated instruments. Washer-disinfectors clean, rinse, and thermally disinfect instruments in a single automated cycle. Cart washers accommodate larger items and cassettes. Load instruments according to manufacturer instructions, typically with hinges open and items positioned to allow water spray to reach all surfaces. Select the appropriate cycle based on soil level and instrument type. The drying cycle is critical for preventing water spots and ensuring instruments are ready for packaging. These machines require regular maintenance, including cleaning spray arms, checking door seals, and verifying cycle parameters. While the initial investment is significant, automated washers improve safety, consistency, and efficiency while freeing staff for other duties.
Special considerations apply to specific instrument types. Lumened instruments like handpieces and air-water syringes have internal channels that require flushing to remove debris. Hinged instruments must be cleaned with hinges fully open. Ultrasonic scaler tips and inserts can be cleaned ultrasonically, but handle them carefully to prevent damage. Burs should be cleaned in specialized bur cleaning devices or carefully by hand with a brush. Throughout cleaning, inspect instruments for damage, wear, or corrosion. Damaged instruments that can't be adequately cleaned or that might injure patients should be repaired or discarded.
After cleaning, verification ensures the process was successful. Visually inspect instruments under good lighting, preferably with magnification. All visible debris should be removed. Protein detection tests using chemical indicators can verify cleaning effectiveness, particularly useful when validating cleaning procedures or training new staff. Any instruments that fail inspection must be recleaned. Document cleaning activities, especially when troubleshooting problems or validating new protocols.
Sterilization Methods and Procedures
Sterilization destroys all forms of microbial life and is the only acceptable processing for critical and most semi-critical dental instruments. Several sterilization methods are available, each with specific advantages and limitations.
Steam sterilization in an autoclave is by far the most common method in dental practices, and for good reason. It's fast, economical, non-toxic, and highly effective against all microorganisms, including bacterial spores. Steam sterilization uses moist heat under pressure to achieve temperatures sufficient to kill microorganisms. The combination of heat, moisture, and pressure provides superior microbicidal activity compared to dry heat at the same temperature. Two basic autoclave types are used in dentistry: gravity displacement and pre-vacuum (dynamic air removal). Gravity displacement autoclaves remove air by displacement as steam enters, with cooler air sinking and exiting through a drain. These require longer exposure times but are less expensive. Pre-vacuum autoclaves actively remove air with a vacuum pump before steam enters, allowing steam to penetrate more rapidly. These offer shorter cycle times and better penetration of porous loads.
Standard sterilization cycles vary by temperature and chamber type. For wrapped instruments in gravity displacement autoclaves, typical parameters are 121°C (250°F) for 30 minutes or 132°C (270°F) for 15 minutes. Pre-vacuum autoclaves typically run 132°C (270°F) for 4 minutes. Times don't include heating and cooling, total cycle time is considerably longer. Unwrapped instruments can be sterilized in shorter cycles, but unwrapped sterilization should only be used for emergencies when wrapped instruments aren't available. The instruments must be used immediately and cannot be stored, as they're unprotected from contamination. Proper chamber loading is essential for success. Don't overload the chamber, which prevents adequate steam circulation. Position packages on their edges with space between them. Allow the drying cycle to complete fully before removing items, as moisture compromises package integrity.
Packaging materials for steam sterilization must be permeable to steam while maintaining sterility after processing. Self-sealing pouches with paper on one side and plastic film on the other are most common for individual instruments or small sets. The paper side allows steam penetration while the plastic side permits contents to be viewed. Place instruments with sharp points toward the paper side to avoid tearing the plastic. Don't overfill pouches, leaving at least one inch from instruments to the seal. Sterilization wraps made of paper or fabric can be used for larger instrument trays and cassettes, typically requiring double-wrapping for adequate sterility maintenance. Cassette systems keep instruments organized and protected but must be specifically designed for steam sterilization with adequate perforations for steam penetration.
Dry heat sterilization uses hot air to kill microorganisms through oxidation. It's appropriate for items that might corrode or rust in steam, such as certain burs and orthodontic instruments, and for items that steam cannot penetrate, like petroleum products and powders. However, dry heat requires much longer exposure times and higher temperatures than steam sterilization. Standard cycles run 160-170°C (320-340°F) for 60-120 minutes in forced air ovens or up to 2 hours in static air ovens. Items must be completely dry before processing and can only be sterilized unwrapped or in special dry heat packaging. The long cycles and high temperatures limit practical use in busy practices. Dry heat is falling out of favor as more instruments are designed for steam sterilization.
Chemical vapor sterilization uses a heated chemical solution of formaldehyde and alcohol under pressure. Operating at 131°C (270°F) for 20-30 minutes, it offers the advantage of not causing corrosion of carbon steel instruments. However, the chemical odor requires excellent ventilation, the vapors can be irritating, instruments must be completely dry before processing, and specific packaging materials are required. This method has declined significantly in dental use as concerns about formaldehyde exposure have grown, and steam sterilization technology has improved.
Low-temperature sterilization methods are designed for heat-sensitive items like plastic devices and certain endoscopes. Options include ethylene oxide gas (rarely used in dental offices due to toxicity and long cycle times), hydrogen peroxide gas plasma, and vaporized hydrogen peroxide. These systems are expensive, have long cycle times (typically 30-75 minutes), and require special packaging and loading procedures. Most dental practices find that very few items genuinely cannot withstand steam sterilization, making the investment in low-temperature equipment unnecessary. When truly heat-sensitive items must be processed, sending them to a facility with appropriate equipment may be more practical than purchasing rarely-used equipment.
Chemical liquid sterilants deserve special mention because they're sometimes confused with sterilization methods. Solutions like glutaraldehyde, ortho-phthalaldehyde, and hydrogen peroxide can achieve sterilization through prolonged immersion times of 6-12 hours. However, instruments processed this way cannot be wrapped, meaning sterility is immediately compromised after removal from solution. There's no way to verify sterilization was achieved, as biological indicators can't be used with liquid immersion. The chemicals are toxic and require significant PPE and ventilation. For all these reasons, chemical sterilization should be considered an absolute last resort only when heat sterilization is truly impossible. The CDC states clearly that heat sterilization is preferred whenever possible and that reusable heat-sensitive items should be replaced with heat-tolerant or disposable alternatives.
Sterilization Monitoring
Sterilization monitoring ensures that your sterilization equipment is functioning correctly and achieving sterilization. A comprehensive monitoring program uses three complementary methods: physical, chemical, and biological.
Physical monitoring should be performed for every single sterilization cycle. Modern autoclaves print out or digitally record time, temperature, and pressure for each cycle. Review these parameters to verify they fall within the acceptable range specified by the equipment manufacturer. The exposure time should meet minimum requirements at the proper temperature, and the pressure should reach the target level. If any parameter is outside acceptable limits, the load is considered non-sterile and must be reprocessed. Physical monitoring catches equipment malfunctions but doesn't guarantee sterilization, as instruments might be improperly packaged or loaded even when cycle parameters are correct.
Chemical indicators change color or display a message when exposed to specific sterilization conditions. External indicators like autoclave tape or labels on packages show that the package has been processed, but don't indicate whether sterilization was achieved. They're useful for distinguishing processed from unprocessed items but have limited value for quality assurance. Internal chemical indicators placed inside packages provide better assurance. Single-parameter indicators respond to one variable (usually temperature), while multi-parameter indicators respond to two or more variables, such as time and temperature or time, temperature, and steam quality. Multi-parameter indicators provide better assurance that all critical sterilization conditions were met. However, even multi-parameter indicators don't guarantee sterilization since they measure physical/chemical conditions, not actual microbial kill. Use chemical indicators with every package and documentthe results in patient records when items are used.
Biological indicators represent the gold standard for sterilization verification because they directly test the sterilization process's ability to kill microorganisms. For steam sterilization, biological indicators contain spores of Geobacillus stearothermophilus, an organism more resistant to heat than pathogens of concern in dentistry. If the sterilization process kills these highly resistant spores, you can be confident it killed less resistant organisms. Run biological indicator tests at least weekly, though some states require more frequent testing. Test every load containing implantable devices before releasing the load for use. Place the biological indicator in the most challenging location in the load, typically the center of the largest package or in a standardized test pack.
Two types of biological indicator systems are available. Mail-in services provide sterilization strips that you process in your sterilizer, then mail to a laboratory for incubation and reading. Results typically take 48 hours. In-office systems use self-contained biological indicators that are incubated in a small in-office incubator with results in 24-48 hours for standard tests or as quickly as 1-3 hours for rapid-readout systems. In-office systems provide faster results and eliminate mailing concerns but require proper incubator maintenance and technique.
If a biological indicator test fails (shows positive for growth), immediate action is required. Take the sterilizer out of service and don't use it until the problem is identified and corrected. Review maintenance records, loading procedures, and operator technique. Run three consecutive biological indicator tests with full loads. Only resume use when all three tests pass. Recall all items processed since the last successful biological test and reprocess them, as sterility cannot be guaranteed. Document the failure, investigation, and corrective actions thoroughly. Positive biological indicators are serious quality assurance failures that indicate a breakdown in the sterilization process, requiring a comprehensive response rather than simply repeating the test.
Beyond routine monitoring, regular equipment maintenance is essential. Follow the manufacturer's service schedule for professional maintenance and calibration. Daily, weekly, and monthly maintenance tasks typically include cleaning door gaskets and the chamber, testing door locks, cleaning air filters, checking for leaks, descaling water systems, and monitoring wear parts. Keep detailed maintenance logs. Equipment that's well-maintained lasts longer, performs more reliably, and provides better sterilization assurance.
Dental Unit Waterlines
Dental unit waterlines present a unique infection control challenge often overlooked until problems occur. The narrow-bore tubing used to deliver water to handpieces, air-water syringes, and ultrasonic scalers creates ideal conditions for biofilm formation.
Biofilm develops when free-floating bacteria attach to the inner walls of water lines and form structured microbial communities embedded in a protective extracellular matrix. This biofilm harbors various microorganisms, potentially including Legionella species, Pseudomonas aeruginosa, and nontuberculous mycobacteria. Water flowing through biofilm-contaminated lines delivers these bacteria directly into patients' mouths and creates aerosols that can be inhaled by both patients and dental staff. Immunocompromised patients face particular risk from waterline-associated microorganisms.
The CDC recommends that dental unit water used for non-surgical procedures should contain no more than 500 colony-forming units per milliliter (CFU/mL) of heterotrophic bacteria, the same standard as EPA drinking water. Untreated dental waterlines commonly contain bacterial counts exceeding 1 million CFU/mL, highlighting the magnitude of the problem. Once biofilm establishes itself, it becomes extremely difficult to remove and continuously seeds the water with bacteria.
Effective dental waterline management requires a comprehensive approach combining multiple strategies. Chemical treatment systems designed specifically for dental waterlines are most commonly used. These involve either connecting the dental unit to an independent water reservoir containing treatment solution or introducing treatment chemicals into municipal water supplies feeding the unit. Various products are available, including chlorine-based systems, hydrogen peroxide, citric acid, and proprietary formulations. Choose products specifically designed and marketed for dental waterline treatment, and follow manufacturer instructions exactly regarding concentration, contact time, and frequency.
Daily protocols form the foundation of waterline management. Before treating the first patient each day, flush each waterline for 20-30 seconds to expel water that stagnated overnight. Between patients, flush lines for 20-30 seconds to physically remove microorganisms that entered from the previous patient's mouth. This brief flushing significantly reduces microbial loads. When using handpieces, ultrasonic scalers, or air-water syringes during treatment, periodically discharge water through the device to flush lines. For surgical procedures, use sterile water or saline as an irrigant rather than water from dental unit lines.
Weekly or monthly maintenance typically involves shock treatment procedures to disrupt biofilm. This might mean running high-concentration disinfectant through lines overnight or during extended periods when the dental unit isn't in use. Follow specific protocols provided by your waterline treatment product manufacturer. Some systems require filter changes at specified intervals.
Periodic water quality testing verifies the effectiveness of your waterline management program. Several commercial laboratories offer testing services where you collect water samples from your dental unit and mail them for bacterial culture. Test at least annually, though quarterly testing provides better quality assurance. If tests reveal bacterial counts above 500 CFU/mL, evaluate your protocols, increase treatment intensity, consult with waterline treatment product manufacturers, and retest after implementing changes. Document all water quality testing results for compliance records.
Air-water syringes deserve special attention in waterline management due to their unique design. When you release the buttons on an air-water syringe, fluid and debris from the patient's mouth can be drawn back into the syringe tip and possibly into the waterline through retraction. Single-use disposable air-water syringe tips eliminate this cross-contamination risk and are strongly recommended. If reusable metal tips are used, they must be removed after each patient and heat sterilized. Anti-retraction valves in dental units help prevent backflow but should not be relied upon as the sole protective measure.
Environmental Infection Control
The dental operatory contains numerous surfaces that become contaminated during patient care and can serve as reservoirs for pathogen transmission. Proper environmental infection control prevents these surfaces from becoming vehicles of cross-contamination.
Dental operatory surfaces fall into two categories requiring different management approaches. Clinical contact surfaces are directly touched during patient care or become contaminated with blood or body fluids. Examples include light handles, chair controls, handpiece holders, bracket tables, unit switches, and drawer handles. These surfaces require either barrier protection or cleaning and disinfection between patients. Housekeeping surfaces like floors, walls, and sinks have minimal hand contact during care and pose a lower infection risk. These require cleaning when visibly soiled and routine periodic cleaning on a regular schedule.
For clinical contact surfaces, you can choose between barrier protection and surface disinfection, or use a combination approach. Barriers include clear plastic wrap, bags, or aluminum foil placed over surfaces before patient care and changed between patients. Barriers work well for surfaces that are difficult to clean effectively, such as light handles with crevices or textured surfaces. They also speed up room turnover between patients. However, you must still clean and disinfect the underlying surface regularly, as barriers can shift during use, may not cover completely, and can develop tears. Remove barriers carefully to avoid contaminating underlying surfaces, discard them as general waste unless visibly bloody, then perform hand hygiene.
Surface disinfection involves applying an EPA-registered hospital disinfectant to clinical contact surfaces between patients. In dental settings, use disinfectants with at least an HIV and HBV claim, though intermediate-level disinfectants with a tuberculocidal claim are preferred. Follow label instructions exactly regarding dilution, contact time (how long the surface must remain wet), and whether the product is suitable for the specific surface material. Some disinfectants can damage certain surfaces with repeated use.
Proper disinfection technique matters as much as product selection. Use a spray-wipe-spray technique: spray the surface, wipe to remove gross contamination, spray again, and allow the surface to remain wet for the full contact time specified on the label. Alternatively, use pre-moistened disinfectant wipes with a wipe-discard-wipe technique, using enough wipes to keep the surface wet for the required contact time. Don't dilute pre-mixed products or use wipes past their expiration date. If surfaces have visible blood or body fluid contamination, clean first with detergent and water, then disinfect.
High-touch surfaces require particular attention during environmental cleaning. In the operatory, these include the dental light handle and switches, chair controls and armrests, handpiece holders, air-water syringe holders, bracket table, countertops, cabinet and drawer handles, faucet handles, computer keyboards and mouse, touchscreens, and phones. Each of these surfaces is touched frequently during patient care and has high potential for pathogen transfer. Don't overlook items outside the immediate treatment area that contaminated hands might touch, such as doorknobs, supply cabinet handles, and equipment adjustment knobs.
Spills of blood or body fluids require immediate attention. Wear appropriate PPE, including utility gloves, a mask, and eye protection. Contain the spill to prevent spread. For large spills, use absorbent material to soak up excess fluid. Clean the area with detergent and water to remove organic material. Apply an EPA-registered hospital disinfectant and allow appropriate contact time. Dispose of contaminated materials as regulated medical waste if saturated with blood. Document any exposure incidents that occur during cleanup.
Aerosol Management
Dental procedures rank among the highest aerosol-generating activities in any healthcare setting, making aerosol management a critical component of infection control that gained heightened attention during the COVID-19 pandemic.
High-speed dental handpieces, ultrasonic scalers, air polishing devices, air-water syringes, and air abrasion units all generate significant aerosols and splatter. Aerosols are tiny particles less than 50 micrometers in diameter that remain suspended in the air for extended periods and can be inhaled deep into the respiratory tract. Splatter consists of larger particles that travel short distances before settling on nearby surfaces. Both contain blood, saliva, and microorganisms from the patient's oral cavity, creating exposure risks for dental personnel and subsequent patients.
The rubber dam, when feasible, provides one of the most effective methods for reducing aerosol generation. Studies show that properly placed rubber dams can reduce airborne particles by up to 70% during procedures. Use rubber dams whenever possible during restorative procedures, endodontic treatment, and other applications where isolation is clinically appropriate. However, rubber dams aren't feasible for all procedures, necessitating other control measures.
High-volume evacuation (HVE) dramatically reduces aerosols when used properly. Position the HVE tip as close as possible to the site of aerosol generation, typically within one inch. The assistant should continuously adjust the HVE position as the operator works to maintain proximity to the source. Proper technique can reduce airborne particles by 90% or more. Ensure vacuum systems provide adequate suction power, maintain equipment regularly to prevent blockages, and train assistants in optimal positioning techniques. Standard saliva ejectors provide minimal aerosol reduction and should not be relied upon as the primary evacuation method during aerosol-generating procedures.
Preprocedural antimicrobial mouth rinses reduce the microbial load in patients' oral cavities before procedures begin. Have patients rinse with chlorhexidine gluconate, essential oil mouthwash, or other antimicrobial rinse for 30-60 seconds before starting treatment. While rinses don't eliminate microorganisms completely and don't reduce viral loads significantly, they provide an additional layer of protection when used as part of a comprehensive approach rather than as a standalone measure.
Ventilation and air quality management became major considerations during COVID-19 and remain important for routine infection control. Ensure HVAC systems are properly maintained with appropriate filter changes. The number of air changes per hour affects how quickly airborne particles are removed. Consider portable HEPA filtration units to supplement building ventilation systems, particularly in treatment areas. Position air purifiers to avoid disrupting aerosol flow patterns toward staff. Some practices have added ultraviolet germicidal irradiation systems, though these require careful installation and maintenance to be effective and safe.
After completing aerosol-generating procedures, allow a settling period before removing PPE and beginning surface disinfection. The time required depends on air exchange rates, typically 15-30 minutes in operatories with standard ventilation. This waiting period lets larger particles settle rather than being disturbed during movement around the room. Use this time for documentation or other tasks outside the operatory.
Handpiece and Dental Device Sterilization
Dental handpieces and attached devices present unique sterilization challenges due to their complex internal structure, moving parts, and tendency for internal contamination through retraction of oral fluids.
When you release pressure on a high-speed handpiece after use, turbine retraction can draw oral fluids back into internal passages. This internal contamination doesn't get removed by external cleaning and creates an infection risk for the next patient. For this reason, handpieces are classified minimally as semi-critical devices requiring heat sterilization between every patient. Chemical disinfection of handpieces is never acceptable, regardless of the chemical used or immersion time.
Proper handpiece processing involves multiple steps. First, run the handpiece for 20-30 seconds to flush internal passages, preferably while pointing into a sink or appropriate collection device. Wipe external surfaces with disinfectant to remove gross contamination. Follow manufacturer instructions for cleaning, which typically involve using enzymatic detergents and paying attention to attachment points and crevices. Thoroughly rinse and dry the handpiece. Lubricate per manufacturer instructions using only approved lubricants; too much lubricant can interfere with sterilization, while too little leads to corrosion and mechanical failure. Package the handpiece in an appropriate sterilization wrap or pouch. Run through a complete sterilization cycle in an autoclave. Many modern handpieces withstand standard steam sterilization cycles, though older models may require special low-temperature cycles.
Automated handpiece maintenance devices simplify lubrication and cleaning, providing consistent results while saving time. These devices typically clean, lubricate, and sometimes purge handpieces in a single cycle. While representing an additional investment, they improve compliance with proper maintenance procedures and extend handpiece life.
Low-speed handpieces, surgical handpieces, and prophylaxis angles all require heat sterilization between patients following similar protocols. Never use cold chemical disinfection or immersion as a shortcut. Disposable prophy angles have become popular precisely because they eliminate sterilization concerns while ensuring a fresh, sterile device for every patient.
Air-water syringe tips, whether metal or disposable, require attention. Disposable tips are strongly recommended and should be used once and discarded. If metal tips are used, they must be removed after each patient and heat sterilized following the same protocols as other critical instruments. The handpiece itself should be cleaned and disinfected, with water flushed through the line between patients.
Ultrasonic scaler tips and inserts are semi-critical devices requiring sterilization. Remove tips from the handpiece, clean manually or ultrasonically, package, and heat-sterilize. Don't forget that the ultrasonic handpiece itself also requires surface disinfection between patients.
Waste Management
Proper dental waste segregation and disposal protect staff, patients, the community, and the environment while ensuring regulatory compliance. Understanding waste categories prevents costly mistakes and potential health hazards.
Regulated medical waste in dental practices includes items saturated or dripping with blood, items that would release blood if compressed, extracted teeth, pathological tissue, and sharps contaminated with patient materials. The definition of what constitutes regulated medical waste varies by state, so check your state regulations. Most dental office waste is actually general waste that can be disposed of through regular trash collection. Over-classification of waste as regulated medical waste increases disposal costs unnecessarily, while under-classification creates compliance problems.
Sharps safety deserves special emphasi,s given the frequency of percutaneous injuries in dental settings. Use puncture-resistant sharps containers that meet OSHA and FDA requirements. Locate containers at the point of use where sharps are generated, not across the room where staff must carry needles. Never overfill containers. Replace them when they reach the fill line, typically about three-quarters full. Never recap needles unless absolutely necessary, and then only using a one-handed scoop technique or a recapping device. Safety-engineered sharps devices with features that shield or retract needles after use should be evaluated and adopted whenever feasible to reduce injury risk.
OSHA requires practices to maintain a sharps injury log documenting every percutaneous exposure incident involving a contaminated sharp. Record the type and brand of device involved, department or work area where exposure occurred, and explanation of how the incident happened. This log helps identify patterns and evaluate safety device effectiveness. Keep the log confidential with no employee names. Maintaining this log is mandatory, not optional, and helps drive continuous improvement in sharps safety.
Biomedical waste requiring special disposal includes regulated medical waste as defined by your state. Collect this waste in red bags or containers clearly labeled with the biohazard symbol. Ensure containers are leak-proof and closable. Store waste in a secure area away from public access. Contract with a licensed medical waste hauler for pickup and proper treatment, either through autoclaving, incineration, or other approved methods. Maintain manifests documenting waste pickups, as these provide your compliance documentation. Never place regulated medical waste in regular trash, down drains, or in any other inappropriate location.
Hazardous waste in dental practices includes amalgam waste, lead foil from X-ray film packets, developer and fixer solutions from film processing, disinfectant chemicals, and certain other materials. Amalgam waste management is particularly important following EPA regulations requiring amalgam separators. Collect all amalgam waste in designated containers specifically for amalgam, never dispose of amalgam down the drain or in regular trash. Contract with a licensed recycler to properly handle collected amalgam. Mercury from dental amalgam remains a significant environmental concern, making proper handling mandatory.
Amalgam separators capture amalgam particles from dental office wastewater before they enter the sewer system. The EPA's 2017 rule requires virtually all dental practices placing or removing amalgam to install ISO 11143-certified amalgam separators, achieving at least 95% amalgam retention. Maintain separators according to manufacturer instructions, replace collection containers before they reach capacity, and maintain documentation of maintenance and waste pickup. Even practices that have gone "mercury-free" and no longer place amalgam may still need separators if they remove existing amalgam restorations.
Waste minimization benefits both the environment and your practice's bottom line. Properly segregate waste to avoid classifying general waste as regulated medical waste, reducing disposal costs. Implement recycling programs for paper, cardboard, and plastics where possible. Use reusable items instead of disposables when clinically appropriate and cost-effective. Train staff on proper waste segregation, as confusion about categories leads to both compliance problems and unnecessary costs.
Bloodborne Pathogen Training and Exposure Control
OSHA's Bloodborne Pathogens Standard protects dental workers from diseases transmitted through contact with blood and other potentially infectious materials. Compliance isn't optional – violations can result in significant penalties and expose practices to liability.
Every dental practice must maintain a written Exposure Control Plan that documents how the practice complies with the Bloodborne Pathogens Standard. This plan must identify job classifications with occupational exposure to blood, explain the methods used to comply with the standard, including engineering controls, work practice controls, PPE, housekeeping procedures, describe the hepatitis B vaccination program, outline post-exposure evaluation and follow-up procedures, explain how the practice communicates hazards to employees, and document employee training. Review and update the plan at least annually and whenever new procedures or equipment affect exposure risk. The plan must be accessible to all employees and OSHA inspectors.
Initial training for all employees with occupational exposure must occur before they perform tasks where exposure might occur, typically during orientation for new hires. Training must cover the bloodborne pathogen standard requirements, disease transmission mechanisms, the exposure control plan and where to access it, recognition of exposure situations, use and limitations of methods to reduce exposure, including engineering controls and PPE, information aboutthe hepatitis B vaccine, actions to take if exposure occurs, reporting procedures, and post-exposure evaluation and follow-up. Annual refresher training is mandatory and must be provided within one year of the previous training. Training must be interactive, allowing employees to ask questions, and must be conducted by a knowledgeable trainer. Simply having employees read materials or watch videos without discussion doesn't satisfy OSHA requirements. Maintain documentation of all training, including dates, content covered, trainer name, and attendee signatures.
Employers must offer hepatitis B vaccination to all employees with occupational exposure to blood within 10 working days of assignment to a position with exposure risk. The vaccine must be provided at no cost to the employee, at a reasonable time and place, and administered by a licensed healthcare professional. If an employee declines vaccination, they must sign a declination form using specific OSHA language indicating they understand the risks and benefits. Employees who decline can change their mind later and receive the vaccination at no cost. Document all vaccinations and declinations carefully.
Despite best prevention efforts, exposure incidents occur when blood or other potentially infectious materials contact employees' mucous membranes, non-intact skin, or penetrate the skin through needlestick or other sharp injuries. When exposure occurs, the immediate response can affect outcomes significantly. Wash wounds and skin with soap and water immediately. Flush mucous membranes with water. Report the incident to a supervisor immediately. Delays in reporting can delay critical post-exposure medical care. Document the incident thoroughly, including how it occurred, what device was involved, what body substances were involved, and what immediate actions were taken.
Post-exposure medical evaluation and follow-up must be provided at no cost to the exposed employee. Evaluation should occur as quickly as possible, ideally within hours. Obtain consent from the source individual for testing when possible, though this isn't always feasible. Test the exposed employee for HIV, HBV, and HCV as appropriate. Based on the source status and exposure type, post-exposure prophylaxis may be recommended, particularly for HIV exposure. Provide counseling about infection risks, interpretation of test results, and necessary precautions during the follow-up period. Schedule follow-up testing at appropriate intervals, typically 6 weeks, 3 months, and 6 months post-exposure. Maintain strict confidentiality of all medical records and test results. The psychological impact of exposure incidents shouldn't be underestimated. Many exposed workers experience significant anxiety even when the infection risk is low.
COVID-19 and Enhanced Infection Control
The COVID-19 pandemic fundamentally changed infection control awareness and practices in dental settings, with many enhanced measures likely to remain permanent additions to standard protocols.
Pre-appointment screening became universal during the pandemic and continues in many practices. Screen patients by phone before appointments, asking about COVID-19 symptoms, recent positive tests, and close contact with infected individuals. Upon arrival, conduct temperature checks and repeat symptom screening. Reschedule symptomatic patients for after their symptoms resolve andthe isolation period ends. While pre-screening was implemented specifically for COVID-19, the concept applies to screening for any communicable diseases and helps protect other patients and staff.
PPE requirements escalated significantly during the pandemic. N95 respirators became standard for aerosol-generating procedures rather than surgical masks. Face shields were added to provide additional barrier protection beyond eye protection alone. Some practices implemented gown changes between every patient rather than wearing the same gown for multiple patients. While supply chains have stabilized and some practices have relaxed to pre-pandemic PPE levels, many continue enhanced protection, recognizing that respiratory pathogens will always pose risks in dental settings.
Waiting room management shifted dramatically to reduce patient density and contact. Practices implemented appointment scheduling with longer intervals to avoid overlap, virtual check-in where patients wait in cars until called, removal of toys, magazines, and other shared items, enhanced cleaning frequency, hand sanitizer stations at entry, and mask requirements for patients when not being treated. Physical distancing became standard, with waiting room seating reduced. Many of these changes improved patient flow and reduced crowding even beyond infection control benefits.
Clinical protocols evolved to minimize aerosol generation and exposure time. Preprocedural antimicrobial mouth rinses became routine before every appointment. Rubber dam use increased for procedures where isolation is possible. High-volume evacuation became universal rather than occasional. Some practices reduced aerosol-generating procedures when alternatives existed. Four-handed dentistry became standard to minimize the number of people in operatories. Treatment sequencing prioritized less aerosol-generating procedures when possible. Appointment duration consideration helped minimize exposure time.
Enhanced air quality measures included improved HVAC maintenance and filtration, portable HEPA air purifiers in operatories, increased ventilation and outside air intake where building systems permitted, and extended settling time after aerosol-generating procedures before room turnover. While these measures were implemented for COVID-19, they improve air quality for all airborne pathogens and chemical vapors.
Looking forward, the pandemic created lasting changes in dental infection control. Patient expectations for visible infection control have increased permanently. Many will never accept a dental provider who doesn't demonstrate serious attention to safety. Respiratory protection awareness among dental workers has increased, and staff are more likely to appropriately use N95 respirators for high-risk situations. Supply chain planning improved, practices maintain better PPE and disinfectant inventories rather than assuming continuous availability. Protocols are more flexible and adaptable – practices demonstrated that they can rapidly implement new measures when threats emerge. The dental profession is better prepared for future infectious disease outbreaks, having developed systems and procedures that can be activated when needed.
Conclusion
Infection control in dentistry represents far more than regulatory compliance. It embodies the profession's fundamental commitment to patient and staff safety. The multi-faceted approach detailed in this guide, from hand hygiene to sterilization monitoring to environmental controls, creates overlapping layers of protection that work together to prevent disease transmission.
Modern dental practices face complex challenges, including bloodborne pathogens, respiratory infections, and environmental contamination, but evidence-based protocols effectively manage these risks when implemented consistently. The key lies not in any single measure but in the comprehensive integration of all components, from proper PPE use to meticulous instrument processing to thoughtful facility design.
Post-pandemic awareness has elevated infection control from a background concern to a visible priority for patients choosing dental providers. Practices that demonstrate transparent commitment to safety protocols, maintain current equipment and supplies, train staff thoroughly and regularly, document procedures comprehensively, and communicate openly with patients about safety measures will build trust and confidence that translates into practice success.
The investment in infection control, whether in time, equipment, supplies, or training, is an investment in the practice's most valuable asset: its reputation for providing safe, quality care. As guidelines evolve and new challenges emerge, successful practices maintain flexibility and commitment to continuous improvement rather than complacency.
For dental professionals, the path forward is clear: treat every patient with appropriate precautions, maintain equipment and supplies proactively, train and retrain staff regularly, monitor and document compliance consistently, stay current with evolving guidelines, and never compromise safety for convenience or efficiency. Patient safety and staff protection are not negotiable aspects of dental practice; they are the foundation upon which everything else is built.
This article is for informational purposes only and does not constitute medical advice. Always consult with qualified healthcare professionals for diagnosis and treatment recommendations specific to your situation.
Frequently Asked Questions
What are the current CDC guidelines for dental infection control?
The CDC's primary guidelines were published in 2003 with updates in 2016 covering standard precautions, PPE, hand hygiene, instrument sterilization, environmental surface disinfection, dental unit waterlines, and special considerations. COVID-19 prompted additional interim guidance emphasizing enhanced PPE, screening, and aerosol management.
How do you properly sterilize dental instruments?
Proper sterilization requires cleaning instruments thoroughly first, packaging in appropriate steam-permeable wraps or pouches, processing in an autoclave at 121°C for 30 minutes or 132°C for 4-15 minutes depending on cycle type, allowing complete drying, and storing sterile packages properly. Monitor with biological indicators weekly.
What PPE is required in a dental office?
At minimum, dental personnel must wear gloves, surgical masks or N95 respirators for aerosol-generating procedures, protective eyewear with side shields or face shields, and protective clothing like long-sleeved gowns or jackets. Requirements vary based on procedure type and anticipated exposure.
How can cross-contamination be prevented in dental settings?
Prevention requires multiple strategies, including hand hygiene before and after patient contact, proper PPE use, instrument sterilization between patients, surface disinfection or barriers on clinical contact surfaces, appropriate waste segregation, proper handling of sharps, and maintaining clean-to-dirty workflow in processing areas.
What is the difference between disinfection and sterilization?
Sterilization destroys all forms of microbial life, including bacterial spores, and is required for critical instruments. Disinfection reduces microorganisms but doesn't eliminate spores. High-level disinfection kills most organisms except high numbers of spores. Intermediate and low-level disinfection are appropriate for surfaces.
How often should autoclave spore testing be performed?
Biological indicator testing should be performed at least weekly in dental practices. Some states require more frequent testing. Test every load containing implantable devices before use. More frequent testing provides better quality assurance and faster detection of equipment problems.
What are standard precautions in dentistry?
Standard precautions assume all patients are potentially infectious and require hand hygiene, appropriate PPE based on anticipated exposure, respiratory hygiene, safe injection practices, proper instrument processing, environmental surface cleaning and disinfection, and appropriate waste management. They apply to all patient encounters regardless of diagnosis.
How do you manage dental waterlines effectively?
Effective management combines daily flushing for 20-30 seconds before the first patient and between patients, chemical treatment with dental waterline products, periodic shock treatments, regular filter changes if applicable, annual water quality testing showing less than 500 CFU/mL bacteria, and using sterile water for surgical procedures.
How has infection control changed after COVID-19?
Changes include enhanced respiratory protection with N95 respirators, pre-appointment patient screening, face shields added to eye protection, improved ventilation and air purification, routine preprocedural mouth rinses, increased rubber dam use, extended time between patients for air exchange, and heightened overall awareness of aerosol risks.